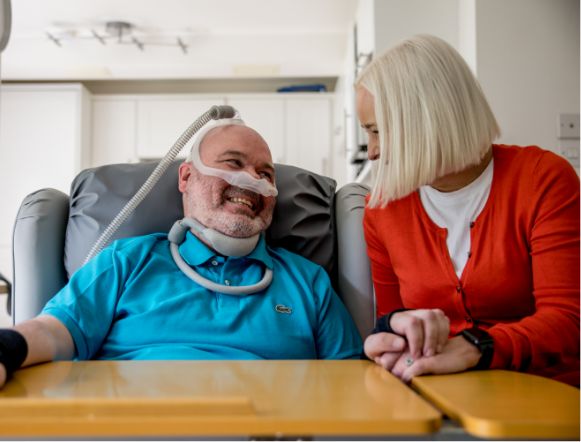
What is MND
Find support
I have MND
I am supporting someone
Get involved
Research
About MND Scotland

The MIROCALS trial was funded largely by EU taxpayers, with further support through donations from several European charities, including a contribution from MND Scotland.
In December 2022, the preliminary findings from the MIROCALS study were announced, which trialed the use of low dose IL-2 for ALS. These early findings were encouraging. However, further research and an analysis of the data was still needed.
Since then, a French company, called ILTOO Pharma, has been selected as the company to commercialise low dose IL2, should the results be sufficiently conclusive. ILTOO is currently also awaiting the finalisation and release of the data by the lead academics on the trial.
In line with our colleagues at the MND Association, at the moment we are unable to influence access to low dose IL-2 for patients with MND as the data needs to be finalised and submitted for publication to allow appropriate review by experts.
To be considered for regulatory review, it must also be presented in the form of a Clinical Study Report (or Regulatory Package) as the data has to be rigorously analysed to determine if the drug is safe and effective.
MND Scotland is a member of the MND Association-convened Treatments Taskforce which is attempting to smooth the path for access as early as possible for people with MND.
Scotland has a separate healthcare system and all new medicines are required to be submitted to the Scottish Medicines Consortium (SMC) – a process that requires publication of the trial data and subsequent submission of a medicine for consideration. The process at SMC invites consultation with patient organisations, such as MND Scotland, and from the public in helping to make their decisions.
MND Scotland is proactively liaising with the relevant organisations in Scotland to ensure the urgent need for new treatments for MND is fully understood and help provide as smooth a path as possible for any treatment that is shown by expert, peer reviewed publication, to be effective in improving quality of life for people with MND.
Sign up
for newsletter
Get the latest news and events straight to your inbox.
You can help create a world without MND
Please consider donating to help make time count for people affected by MND.