What is MND
Find support
I have MND
I am supporting someone
Get involved
Research
About MND Scotland

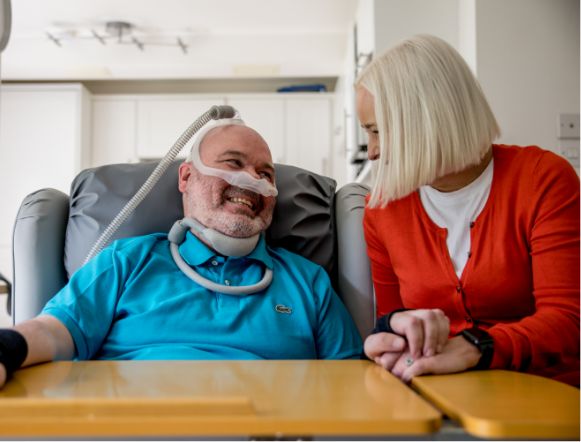
We have now been told by the Repurposing Team they won’t be able to make a decision until the results of the MIROCALS trial (which tested interleukin-2 as a potential treatment for MND) have been published in a peer-reviewed journal.
They have made it clear that while they have gathered information from a number of stakeholders, it’s not possible to properly assess the potential benefits and risks of low dose aldesleukin without the trial results.
The submission has now been placed on the repurposing programme’s monitor list for review once the trial results are published.
Following this update from the Repurposing Team, we have again been in touch with the lead investigators in the MIROCALS trial to ask, once more, when they expect the results of the MIROCALS trial to be published.
We have also this week written once again to ILTOO Pharma, urging them to respond to our letter of 12 October seeking clarification about the managed access programme.
Our focus as MND charities remains on working together to secure equitable access for people with MND to safe, effective and proven treatments at the earliest opportunity.
Sign up
for newsletter
Get the latest news and events straight to your inbox.
You can help create a world without MND
Please consider donating to help make time count for people affected by MND.